Description
Prevent, treat and destroy biofilms by targeting the immune system

Tammy Kielian, PhD
More than a million cases of hospital-acquired infections can be traced to biofilm, a slimy coating of microbes that can form on medical devices and implants.
Biofilm infections are notoriously difficult to treat without removing or replacing the infected medical device. Treatment is further hindered by the development of antibacterial resistant strains of microbes.
But researchers at the University of Nebraska Medical Center have discovered a new way to combat device-related infections. Rather than pursue traditional routes of treatment with antibiotics, Dr. Tammy Kielian has discovered a method to prevent, treat and destroy biofilms that adhere to implanted medical devices.
Dr. Tammy Kielian has discovered that the inhibition of a certain population of cells known as myeloid-derived suppressor cells, enhances the ability of the body’s immune system to attack and clear biofilms.
By targeting a patient’s own cells, rather than using antibiotics, this approach may also help decrease the likelihood of developing antibiotic-resistant bacteria.
To discuss licensing opportunities please contact Matt Boehm, PhD, at mboehm@unmc.edu or 402-536-9881.
Technical details
Targeting myeloid-derived suppressor cells (MDSCs) for the prevention/treatment of bacterial biofilm infections
Dr. Kielian has generated a method to enhance the ability of a patients’ immune system (specifically monocytes and macrophages) to eliminate bacterial biofilms. This method focuses on the inhibition of a specific cell populations know as myeloid-derived suppressor cells (MDSCs). Using a mouse model of S. aureus orthopedic device infection, Dr. Kielian has shown that MDSCs are the main cellular infiltrate associated with S. aureus orthopedic biofilm infections.
In the presence of MDSCs, inflammation persists but does not facilitate bacterial clearance. In addition, monocyte/macrophage infiltrates are reduced and T cell proliferation and cytokine production is inhibited. Depletion of MDSCs using an anti-Ly6G antibody enhanced the proinflammatory properties of infiltrating monocytes and macrophages allowing for improved bacterial clearance. These data demonstrate the effectiveness of targeting and inhibiting MDSCs for the treatment of medical device associated biofilms.
We are interested in collaborating with an industry partner to test existing drugs capable of inhibiting MDSCs or developing new therapeutics to inhibit the function of MDSCs for the treatment of biofilm infections.
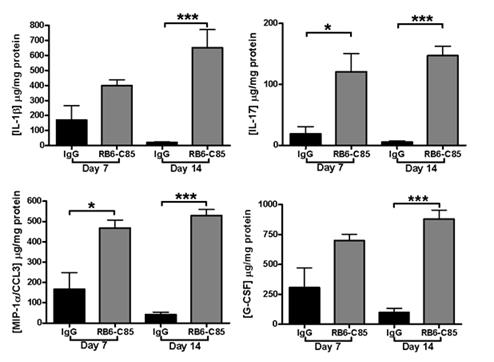
Levels of inflammatory mediators following inhibition of MDSCs. Inhibition of MDSCs leads to significantly heightened inflammatory mediators.