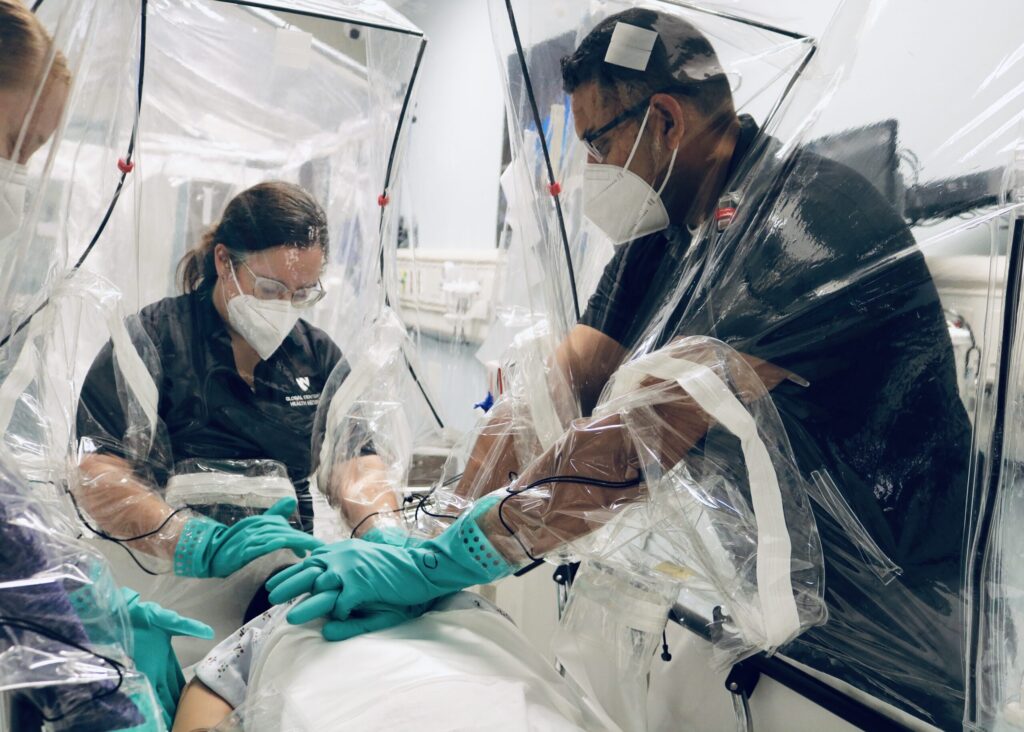
Clinical staff at UNMC/Nebraska Medicine perform life-saving procedures during a medical simulation using an early prototype of the Carecubes isolation unit.
OMAHA, Nebraska (March 31, 2026)—The last several months have been a whirlwind for a device developed at UNMC’s world-class infectious diseases division.
Carcubes quietly launched its innovative product in several U.S. hospitals in 2025, and was prominently featured an October episode of NBC’s hit medical drama, Chicago Med. More recently, Carecubes announced a successful $6.5 million Series A funding round.
The product’s commercial growth and notoriety represent a major milestone for a life-saving device more than 10 years in the making.
Originally developed in response to the 2014 Ebola outbreak in Sierra Leone, today’s Carecube is a transportable and easily assembled mobile isolation unit for patients suffering from contagious diseases. In wrapping the patient in protective gear, the Carecube saves healthcare workers time and energy when donning or doffing personal protective equipment during an outbreak.
“I’ve been in our emergency room, assessing suspect patients or potential patients, possibly with high consequence infectious diseases and wished we had this device available,” said James Lawler, the Associate Director for International Program and Innovation at UNMC’s Global Center for Health Security and the Deputy Director of the Nebraska Biocontainment Unit.
Lawler, in effect, granted his own wish, as he and his team of world renown infectious disease experts from UNMC helped develop the isolation unit, including Mara Jana Broadhurst, MD, PhD; David Brett-Major, MD; and Chris Kratochvil, MD.
Now he and virtually any other clinical facility on the planet have access to a far better and simpler way to protect patients, providers and the communities they serve from infectious diseases—and perhaps even more readily contain future outbreaks.
UNMC’s infectious disease bona fides gained wide notoriety during the 2014 Ebola outbreak. Three patients were medically evacuated to UNMC’s biocontainment unit during what was the worst Ebola outbreak in history.
The hemorrhagic fever swept through Sierra Leone and five other African countries before landing in Europe and even the United States. More than 28,600 people in 10 countries were infected, leading to 11,325 deaths, 221 of which were healthcare workers.
As the pandemic raged in Sierra Leone, the U.S. Department of Defense’s advanced research projects agency, or DARPA, initiated a new program in response. The goal of the P3 Program was to accelerate the innovation and development process for tools and solutions that might help fight or prevent infectious disease outbreaks.
Otherlab, an independent research and development firm in San Francisco, answered the call and started work on a novel solution. The idea, first put forward by Otherlab founder, Saul Griffith, was to wrap the patient in PPE rather than the clinical workers.
That idea became the ISTARI device, or the Isolation System for Treatment and Agile Response for High-Risk Infections. It was the earliest version of today’s Carecube.
Once the Ebola pandemic ended, despite its promise and potential, Griffith’s idea landed on a dusty shelf, seemingly forgotten.
Several years later, in 2018—as Lawler and so many others fought a new Ebola outbreak, this time in the Democratic Republic of the Congo—he recalled Griffith’s innovative idea.
In the small world of the infectious disease community, Lawler knew some of the people that worked on the ISTARI project. He made a few calls.
“They connected me to Saul,” Lawler said. “I always had that project in the back of my mind, and how we might reinvent the wheel for the Congo outbreak.”
As the regional Ebola outbreak continued, the rising threat of a novel coronavirus added urgency to the revived ISTARI project.
The COVID-19 pandemic exposed unexpected vulnerabilities in the supply chain. Things like masks, gloves, swabs and other important protective gear clinical workers needed in testing and treating COVID patients were soon in short supply or just unavailable.
Suddenly, Carecubes’ approach—wrapping the patient in protective gear and potentially turning any hospital room in an isolation unit—attracted wide and robust interest.
By early 2020, the Otherlab-UNMC collaboration created a startup company around the device. Griffith called his friend, Alex Laskey, to act as CEO.
In April 2020 Laskey’s brother, a clinical psychiatrist, was primarily treating fellow clinicians suffering from PTSD caused by conditions the pandemic created.
“I thought to myself in April 2020, watching nurses and doctors being treated as disposable commodities, ‘Who am I to say no,’” he said.
Five years later, Carecubes has an FDA-approved device and is actively marketing their products.
“I’m really proud of the device we created, and I’m excited to see it help people,” Lawler said. “It’s been a great collaboration process. I think everybody can point to a feature that was their idea…And some are like, ‘Why hasn’t that been done before?’ Just brilliant ideas.”
The Carecube cleared the FDA in early 2024 and is available in six major U.S. hospitals today. Manufactured in Minnesota, Carecube units began arriving in additional half-dozen hospitals before the end of summer in 2025.
Made to travel the globe for quick and easy deployment anywhere—whether a small clinic in rural Nebraska or an improvised encampment in one of the most remote areas on the planet—the value of the Carecube device is not limited to notoriously exotic diseases like Ebola.
Isolating measle patients or cases of drug-resistant tuberculosis are but two examples where isolation capability would be necessary at a remote facility not typically equipped to handle highly infectious diseases.
A recent study showed that just 2.4 percent of hospitals—and zero senior nursing homes—have airborne infection isolation rooms.
“We are incredibly proud of our entire team,” Lawler said, “which has been instrumental in getting these devices to this point and continues to validate UNMC’s standing as one of the world’s leading institutions for managing high consequence infectious diseases.”
Read article

 Blake is expected to deliver an update on his company’s progress, which has been preparing to clear the FDA and launch a product that began as a UNMC innovation. The innovation is a one-handed syringe invented by a former pediatric ophthalmologist who wanted more precision and agility when injecting medications into his patients’ eyes.
Blake is expected to deliver an update on his company’s progress, which has been preparing to clear the FDA and launch a product that began as a UNMC innovation. The innovation is a one-handed syringe invented by a former pediatric ophthalmologist who wanted more precision and agility when injecting medications into his patients’ eyes.

 The Boot Camp focuses on several key areas relevant to a successful career in technology transfer, including:
The Boot Camp focuses on several key areas relevant to a successful career in technology transfer, including:


 Steel Works is focused on helping early-stage startups and founders working with “healthtech” innovations from the University and the greater Nebraska community. In collaboration with the health venture company CQuence Health, Steel Works mentors and advises a small cohort of startups as they work to build their investor networks, business models and company messaging.
Steel Works is focused on helping early-stage startups and founders working with “healthtech” innovations from the University and the greater Nebraska community. In collaboration with the health venture company CQuence Health, Steel Works mentors and advises a small cohort of startups as they work to build their investor networks, business models and company messaging.


 The University of Nebraska system landed No. 57 among the National Academy of Inventors’ national
The University of Nebraska system landed No. 57 among the National Academy of Inventors’ national 





 “Think of our office as the front door that bridges the university with industry,” she told the gathering during a 15-minute presentation. “And that goes both ways.”
“Think of our office as the front door that bridges the university with industry,” she told the gathering during a 15-minute presentation. “And that goes both ways.”
 “HemaGlobal’s rapid growth was made possible by the vital support of the
“HemaGlobal’s rapid growth was made possible by the vital support of the  OMAHA, Nebraska (February 23, 2026)—Impower Health, Inc., a startup company built on an innovation developed at the University of Nebraska at Omaha, recently secured an undisclosed investment from Boomerang Ventures while adding a new CEO.
OMAHA, Nebraska (February 23, 2026)—Impower Health, Inc., a startup company built on an innovation developed at the University of Nebraska at Omaha, recently secured an undisclosed investment from Boomerang Ventures while adding a new CEO.

 The honor was part of the TechPipeline Impact Awards, which recognizes the “exceptional individuals who elevate the field of technology transfer through their dedication, innovation, and expertise.” TechPipeline is one of the leading technology transfer industry organizations in the United States, providing guidance, training and best practices standards.
The honor was part of the TechPipeline Impact Awards, which recognizes the “exceptional individuals who elevate the field of technology transfer through their dedication, innovation, and expertise.” TechPipeline is one of the leading technology transfer industry organizations in the United States, providing guidance, training and best practices standards.




