Sam Sanderson, PhD
OMAHA, Neb. (Oct. 5, 2016)—Prommune Inc. was recently awarded a federal contract from the National Institute of Allergy and Infectious Diseases (NIAID) for the development of vaccines to Toxoplasma gondii infections.
The two-year Phase I Small Business Innovation Research contract for $438,932 was awarded by NIAID (contract #HHSN272201600038C) to Prommune, an Omaha startup that spun out of research from Sam Sanderson, PhD, a research associate professor in the Department of Pharmaceutical Sciences at UNMC.
Dr. Sanderson founded Prommune around an immune stimulating peptide he and his team created. Early tests show the peptide, called EP67, has the ability to enhance a more robust natural immune response against infections.
“EP67 is a platform technology,” said Prommune CEO Sam Al-Murrani, PhD, “but with this contract we’ll begin to see the wider therapeutic potential of EP67.”
Prommune and Dr. Sanderson will partner with University of Nebraska at Omaha biology professor and T. gondii expert Paul Davis, PhD, to develop a possible vaccine against T. gondii infections.
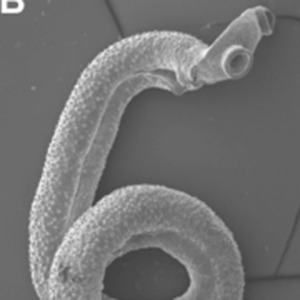
An estimated 60 million in the U.S. are infected with T. gondii parasites, which is typically acquired from infected cats or eating undercooked meat. For most people, the infection goes unnoticed or presents only temporary flu-like symptoms. Healthy immune systems usually prevent the parasite from causing serious illness, according the Centers for Disease Control and Prevention website. But for people with weak or compromised immune systems an infection could lead to a serious condition called toxoplasmosis.
Toxoplasmosis occurs when the parasites create cysts within the body. The condition is particularly dangerous if the parasites end up in the brain where cysts can cause neurological and behavioral disorders, or even death. For pregnant women, a T. gondii infection could be transmitted directly to the fetus, creating an increased potential for birth defects. Contact with cat feces is one of the most common routes of transmission, which is why pregnant women are strongly advised against handling cat litter boxes.
Drs. Sanderson and Davis will look to prevent T. gondii infections using the EP67 peptide in the development of vaccines in the Phase I proof-of-concept round of testing. If successful, researchers will apply for Phase II, which would involve further process development leading to human clinical trials and FDA approval.
This project has been funded in whole or in part with Federal funds from the USC 2304(b)(2) USC 253(b)(2), National Institutes of Health, Department of Health and Human Services, under Contract No. HHSN272201600038C.